Development of An Asian-Skin In A Microfluidic Plate for Safety Assessment
Key Information
AM Tech
Bioprinting Industry
Cosmetic Potential Applications
- Translating manual fabrication of in vitro human skin models to bioprinted models overcomes limitations in terms of labour and time-intensive process
- F4Application of bioprinted skin tissue with Asian origin opens is directed at Asian market that differs with Caucasian skin due to different diet, environment and air pollution, low UV exposure etc
Collaborators
SC3DP (NTU) X Denova Sciences Problem Statement
Fabrication of in vitro human skin models generally involves multiple steps using the manual method. Bioprinting has shown the potential to replace labour and time intensive processes in skin model fabrication. However, commercially available models such as Epiderm are difficult to scale because the seeding and culturing protocols were optimised for standard cell culture plates. In addition, these standard cell culture plates are generally not of Asian origin and hence unable to provide a total solution to the Asian market. Asian skin is different from Caucasian skin due to different diets, environment and air pollution, low UV exposure etc.
Objective
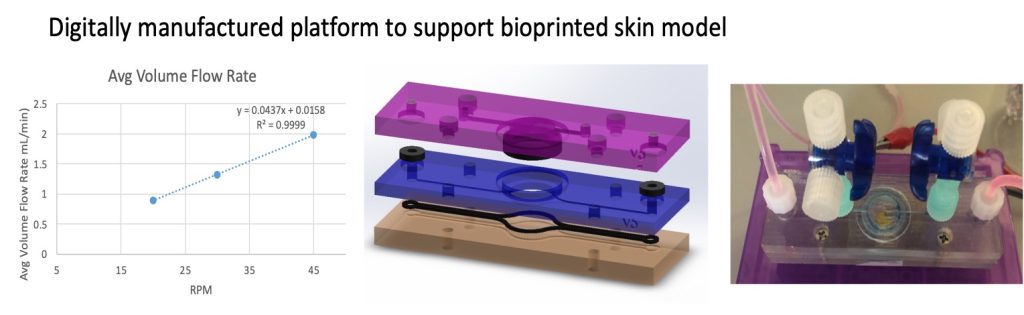
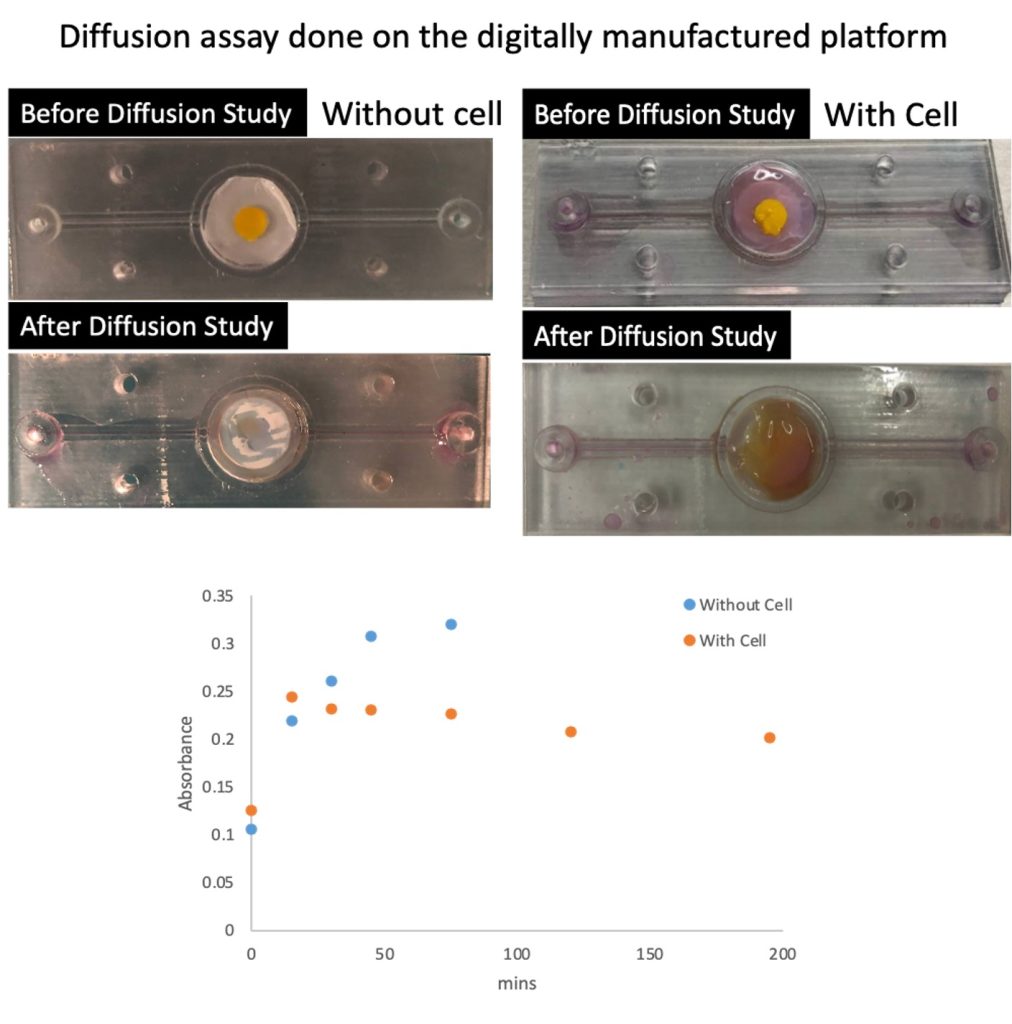
- To develop an Asian skin-on-a-plate device capable of a range of capabilities such as skin maintenance in a viable and productive state, control and modulation of flow environment, allowing observation of sample condition in situ, and enabling the easy selective collection of samples for future analysis.
- The skin cells used in the culture should be closely related to the targeted consumer population (Asian market).
- 3D bioprinting of skin model, with close resemblance to the biochemical and physiological properties of human skin.
Key Benefits/Outcomes
- Developed printing process and established printing parameters of in vitro skin model using both extrusion and microvalve bioprinting techniques, for rapid fabrication of full-thickness human skin model.
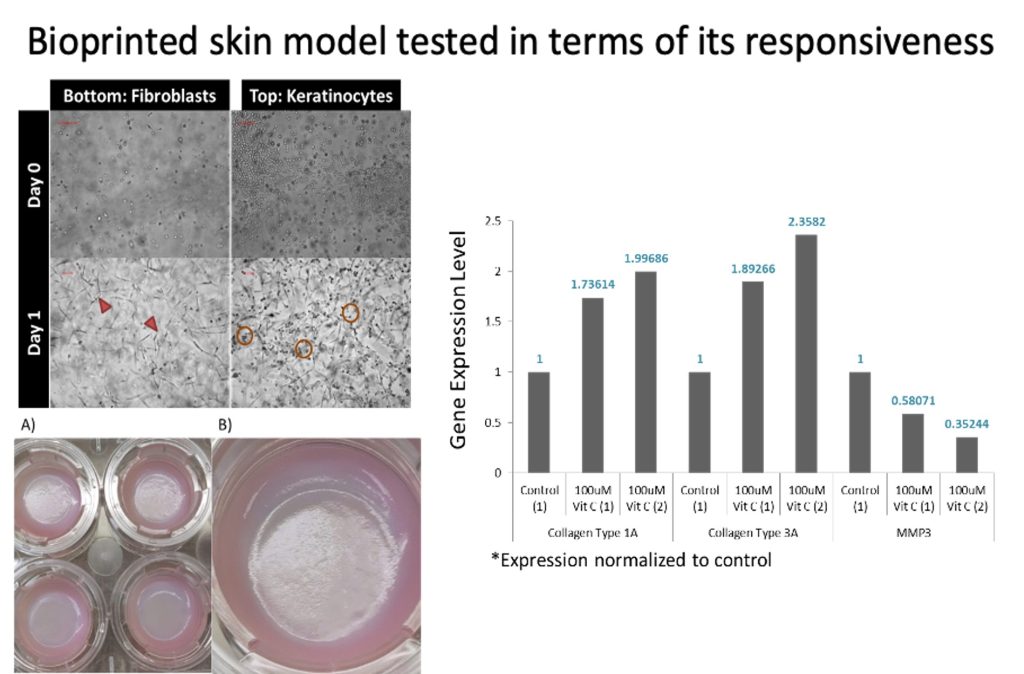
- Demonstrated functionality of bioprinted skin model using Vitamin C as the chemical reagent. The bioprinted model sees an upregulation in Collagen Type 1A, Collagen Type 2A and downregulation MMP 2 in response to Vit C exposure.
- Patent applications on i) bioprinting processes for fabricating skin model, and ii) a digitally manufactured platform to maintain skin model, modulate media flow, and allowed collection of eluent for analysis.